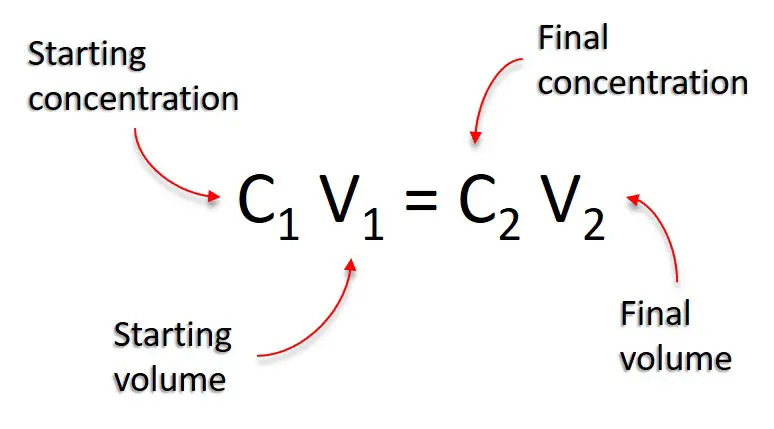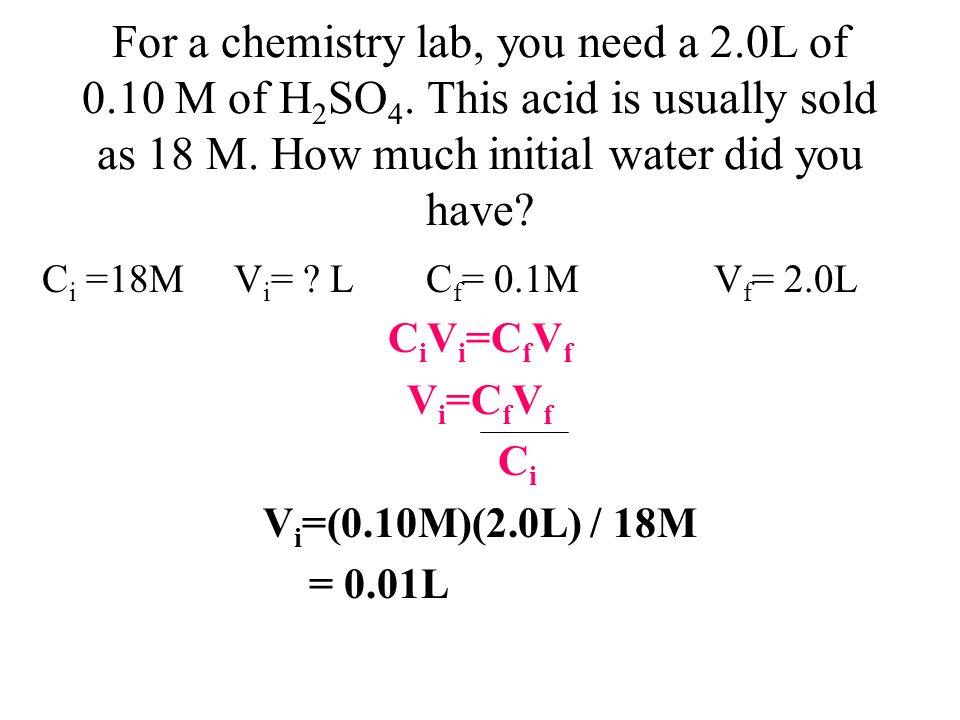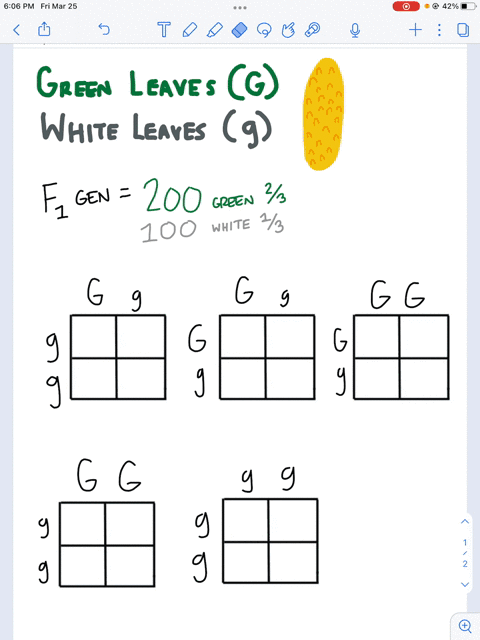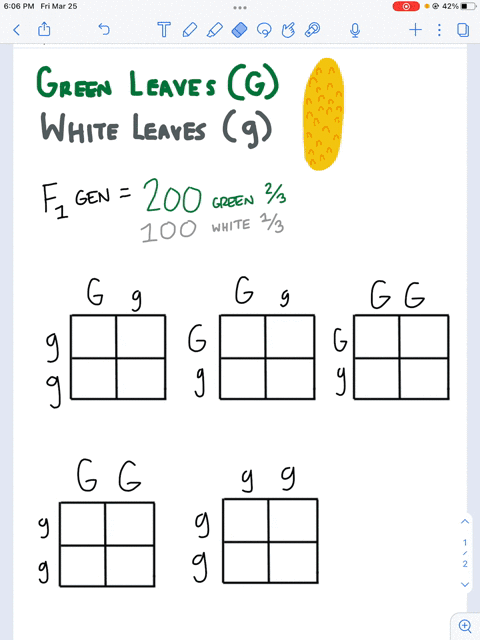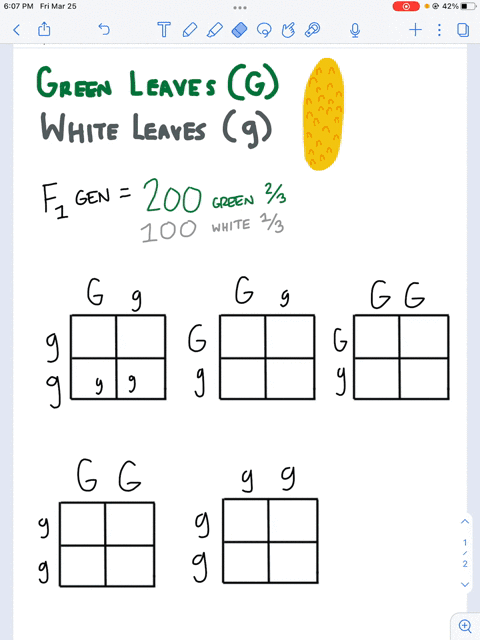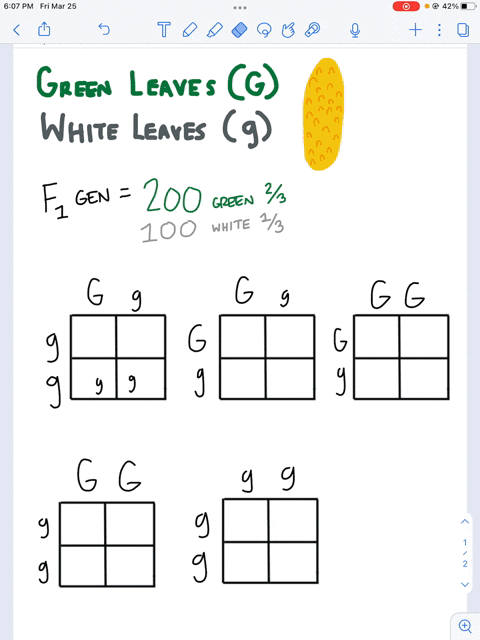
SOLVED: Questions Complete the table below and show a sample of your calculations. You will be using the formula CiVi = CfVf (equivalent to C1V1 = C2V2). The initial maltose concentration (Ci)
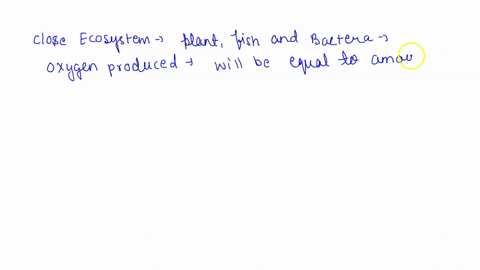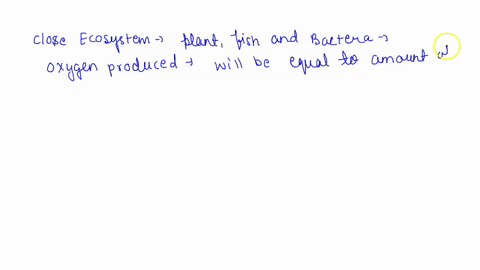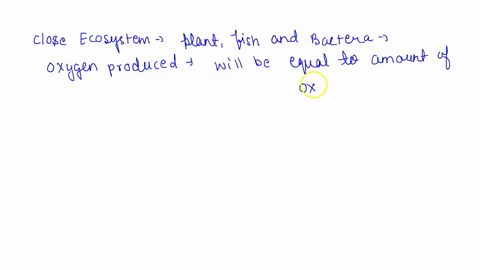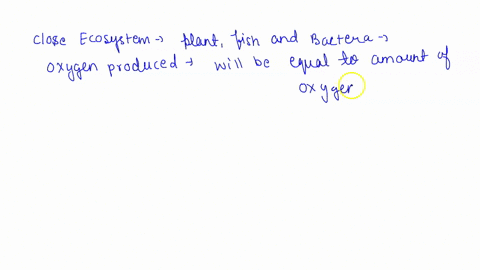
SOLVED: Questions Complete the table below and show a sample of your calculations. You will be using the formula CiVi = CfVf (equivalent to C1V1 = C2V2). The initial maltose concentration (Ci)